New 2016 product catalogue
PurePro has launched its new website. The website features all of our products listed in our new product catalog and features a comprehensive technical support area.
[ what is reverse osmosis ] [ RO advanced water treatment ] [ RO Renaissance ] [ water from source to tap ] [ water quality problem ] [ treating the water ] [ what make water hard ] [ why is RO better ] [ Japan Nuclear Crisis Affect Drinking Water ] [ 3 Great Reasons To Remineralize Your Pure Water ]
What Is. . . Reverse Osmosis
Anyone who has been through a high school science class will likely be familiar with the term osmosis. The process was first described by a French Scientist in 1748, who noted that water spontaneously diffused through a pig bladder membrane into alcohol. Over 200 years later, a modification of this process known as reverse osmosis allows people throughout the world to affordably convert undesirable water into water that is virtually free of health or aesthetic contaminants. Reverse osmosis systems can be found providing treated water from the kitchen counter in a private residence to installations used in manned spacecraft.
Reverse Osmosis is a technology that is found virtually anywhere pure water is needed; common uses include:
Drinking Water
Humidification
Ice-Making
Car Wash Water Reclamation
Rinse Waters
Biomedical Applications
Laboratory Applications
Photography
Pharmaceutical Production
Kidney Dialysis
Water used in chemical processes
Cosmetics
Animal Feed
Hatcheries
Restaurants
Greenhouses
Metal Plating Applications
Wastewater Treatment
Boiler Water
Battery Water
Semiconductor production
Hemodialysis
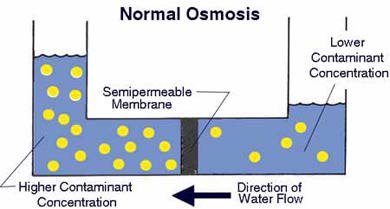
How Reverse Osmosis Works
A semipermeable membrane, like the membrane of a
cell wall or a bladder, is selective about what it
allows to pass through, and what it prevents from
passing. These membranes in general pass water very
easily because of its small molecular size; but also
prevent many other contaminants from passing by
trapping them. Water will typically be present on both
sides of the membrane, with each side having a
different concentration of dissolved minerals. Since
the water i the less concentrated solution seeks to
dilute the more concentrated solution, water will pass
through the membrane from the lower concentration side
to the greater concentration side. Eventually, osmotic
pressure (seen in the diagram below as the pressure
created by the difference in water levels) will
counter the diffusion process exactly, and an
equilibrium will form.
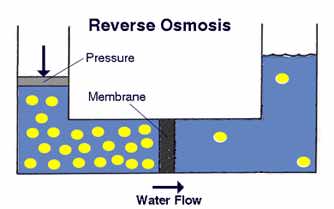
The process of reverse osmosis forces water with a greater concentration of contaminants (the source water) into a tank containing water with an extremely low concentration of contaminants (the processed water). High water pressure on the source side is used to "reverse" the natural osmotic process, with the semi-permeable membrane still permitting the passage of water while rejecting most of the other contaminants. The specific process through which this occurs is called ion exclusion, in which a concentration of ions at the membrane surface from a barrier that allows other water molecules to pass through while excluding other substances.
Semipermeable membranes have come a long way from the
natural pig bladders used in the earlier osmosis
experiments. Before the 1960's, these membranes were too
inefficient, expensive, and unreliable for practical
applications outside the laboratory. Modern advances in
synthetic materials have generally solved these
problems, allowing membranes to become highly efficient
at rejecting contaminants, and making them tough enough
to withstand the greater pressures necessary for
efficient operation.
Even with these advances, the "reject" water on the
source side of a Reverse Osmosis (RO) system must be
periodically flushed in order to keep it from becoming
so concentrated that it forms a scale on the membrane
itself. RO systems also typically require a carbon
prefilter for the reduction of chlorine, which can
damage an RO membrane; and a sediment prefilter is
always required to ensure that fine suspended materials
in the source water do not permanently clog the
membrane. Hardness reduction, either through the use of
water softening for residential units or chemical
softening for industrial use, may also be desirable in
hard water areas.
Low Pressure (Residential) Systems
Low pressure RO systems generally refer to those systems
with a water feed pressure of less than 100 psig. These
are the typical countertop or undersink residential
systems that rely primarily on the natural water
pressure to make the reverse osmosis process function; a
typical system is shown schematically below.
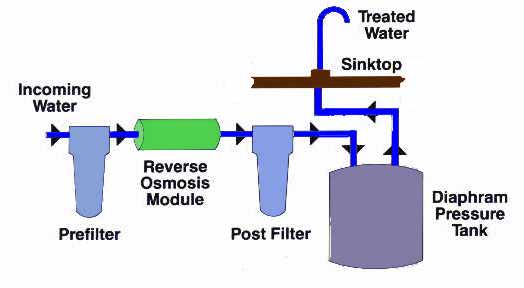
Typical Point of Use Reverse Osmosis System
Countertop units typically have an unpressurized storage
tank; Undersink units typically have a pressurized
accumulator storage tank where the water pressure tends
to increase as the tank fills. This pressurized system
provides sufficient pressure to move the water from the
undersink storage tank to the faucet. Unfortunately,
this also creates a back pressure against the membrane,
which can decrease its efficiency. Some units overcome
this by using unpressurized tanks with a pump to get the
treated water where it is needed.
Low pressure units typically provide between 24 and 35
gallons per day of water (Pure-Pro System offer 50-80
gallons per day), Water purity can be as high as 95
percent of rejection. These systems can be highly
affordable, undersink units starting at about US $500.
These units produce water for a cost as low as five
cents per gallon once maintenance and water costs are
factored in. Maintenance usually requires replacing any
pre- or postfilters (typically one to four times per
year); and the reverse osmosis cartridge once every two
to three years, depending on usage. Look for PPW Home
Reverse Osmosis System to find products that have been
successfully tested to industry performance standards;
and to Certified Water Specialists (CWS I-V), Certified
Sales Representatives (CSR), and Certified Installers
(CI) for advice on your water needs, and equipment
installation.
High Pressure (Commercial/Industrial) Systems
High pressure systems typically operate at pressures
between 100 and 1000 psig, depending on the membranes
chosen and the water being treated. These systems are
usually used in industrial or commercial applications
where large volumes of treated water are required at a
high level of purity.
Most commercial and industrial systems use multiple
membranes arranged in parallel to provide the required
quantity of water. The processed water from the first
stage of treatment can then be passed through additional
membrane modules to achieve greater levels of treatment
for the finished water. The reject water can also be
directed into successive membrane modules for greater
efficiency (see diagram below), though flushing will
still be required when concentrations reach a level
where fouling is likely to occur.
High pressure industrial units typically provide from 10 gallons to thousands of gallons per day of water with an efficiency of 1-9 gallons of reject water per gallon of treated water. Water purity can be as high as 95 percent. These systems tend to be larger and more complicated than low pressure systems, and this is reflected in their costs, which range from US $1000 through tens of thousands of dollars for a large, multi-module unit capable of providing desalinated drinking water for a resort facility or water bottling plant.
What Reverse Osmosis Treats
Reverse osmosis can treat for a wide variety of health
and aesthetic contaminants. Effectively designed, RO
equipment can treat for a wide variety of aesthetic
contaminants that cause unpleasant taste, color, and
odor problems like a salty or soda taste caused by
chlorides or sulfates.
RO can also be effective for treating health
contaminants like arsenic, asbestos, atrazine
(herbicides/pesticides). fluoride, lead, mercury,
nitrate, and radium. When using appropriate carbon
prefiltering (commonly included with most RO systems),
additional treatment can also be provided for such
"volatile" contaminants as benzene, trichloroethylene,
trihalomethanes, and radon. Some RO equipment is also
capable of treating for biological contaminants like
Cryptosporidium. The Water Quality Association (WQA)
cautions, however, that while RO membranes typically
remove virtually all known microorganisms and most other
health contaminants, design consderations may prevent a
unit from offering foolproof protection when
incorporated into a consumer drinking water system.
When looking for a product to treat for a given health
contaminant, care should be used to find products that
have been tested successfully for such purposes at a
quality testing laboratory.
Conclusion
Reverse osmosis is a relatively new, but very effective,
application of an established scientific process.
Whether it is used to meet the needs of a typical family
of four, or the needs of an industrial operation
requiring thousands of gallons per day, it can be a cost
effective to provide the required quantity of highly
treated water. With continual advances in system and
membrane design that boost efficiency and reliability,
RO can be expected to play a major role in water
treatment for years to come.

